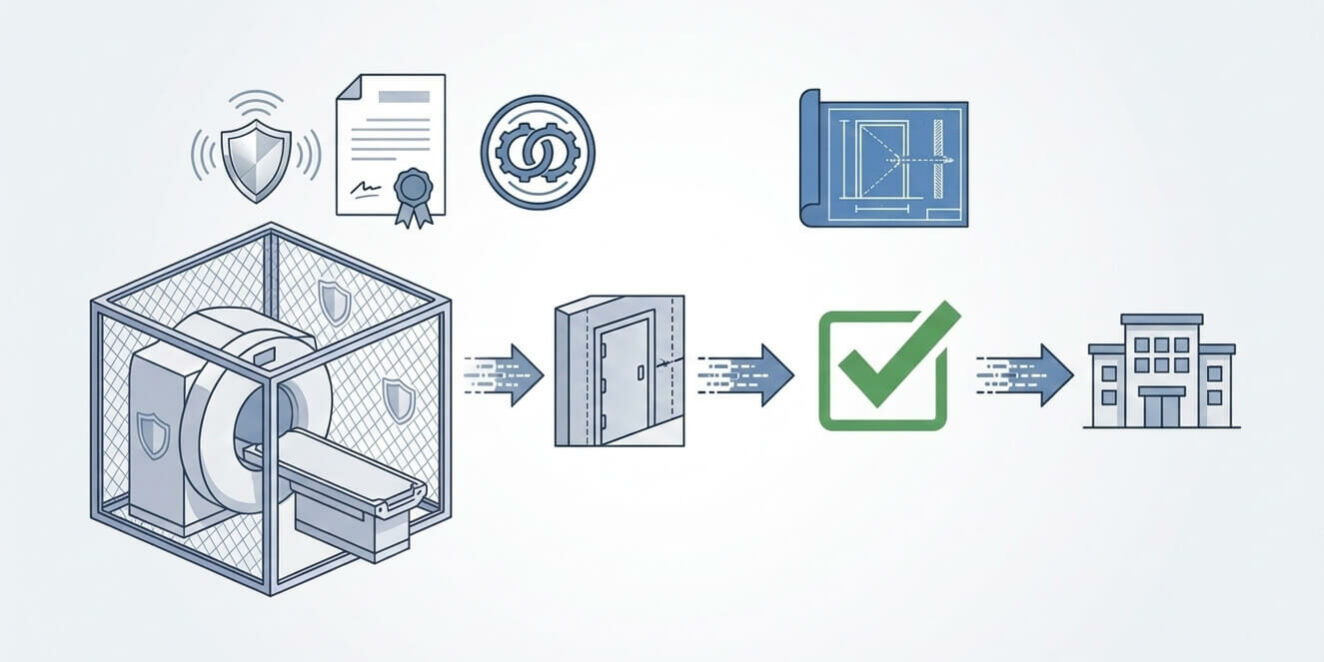
In the U.S. healthcare market, MRI shielding certification is more than a technical formality; it is a critical component of risk management and facility accreditation. Whether you are aiming for ACR (American College of Radiology) accreditation or preparing for a Joint Commission inspection, the certification of your RF enclosure provides the only objective proof that your facility meets the stringent requirements of magnet OEMs (GE, Siemens, Philips).
1. Adherence to IEEE-299 and MIL-STD standards
The first question for any U.S. supplier must concern their testing methodology. In the United States, professional shielding certification must follow the IEEE-299 or MIL-STD-285 standards. These protocols must be specifically tuned to the Larmor frequencies of the magnet—typically 63.8 MHz for 1.5T or 127.7 MHz for 3T systems. Ensure the final report includes attenuation data for both electric and magnetic fields to guarantee full compliance.
2. Credentials and equipment calibration
A rigorous certification protocol is only as reliable as the tools used. U.S. clients should demand proof that the testing technician is qualified and that their instrumentation, such as Signal Generators and Spectrum Analyzers, carries up-to-date NIST-traceable calibration certificates. Understanding how to choose the right MRI shielding partner in the U.S. market involves verifying their history of successful inspections by hospital physicists and safety officers.
3. Verification of ACR Zone IV integrity
The certification must go beyond testing the center of a panel. It must validate every penetration point where RF leakage could compromise the safety of the ACR-defined Zone IV. A professional protocol includes specific leakage tests on:
* RF Door Assemblies: Testing the fingerstock gaskets under mechanical load.
* Filter Banks and Penetration Panels: Verifying the attenuation of power filters and medical gas manifolds.
* HVAC Waveguides: Ensuring honeycomb filters are electromagnetically bonded to the shield to prevent external interference from building systems.
4. Remediation and “Right to Cure”
From a contractual standpoint, ensure your supplier provides a clear remediation path. If a “leak” is detected during the commissioning phase, the supplier should be responsible for immediate diagnosis, repair, and a documented re-test at no additional cost before the “As-Built” documentation is signed off.
5. Comprehensive documentation for accreditation
Upon completion, you must receive a detailed “Shielding Effectiveness Report.” This document is vital for your site’s physics survey and future magnet upgrades. It should include a map of test points, decibel (dB) attenuation values, and a statement of compliance. In conclusion, demanding a transparent certification protocol is the best way to protect your clinical investment and ensure the highest diagnostic reliability for your patients.
Need help or have a project in mind?
Share the details with us! Our experts are at your disposal and ready to support you every step of the way, showing you the best solution for your needs.
Call us at +1 646 372 2191 or write us at info@qualityshielding.com